This web page was produced as an assignment for Genetics 564, an undergraduate course at UW-Madison .
What is a protein domain?_____________________________________________________________________________________
Domains are conserved structural or functional units of a protein. They may exist independently of their protein chain and usually contribute to the overall function of the protein by being part of specific interactions [1].
Domains are conserved structural or functional units of a protein. They may exist independently of their protein chain and usually contribute to the overall function of the protein by being part of specific interactions [1].
|
DRD2 domain structure____________________________________________________________
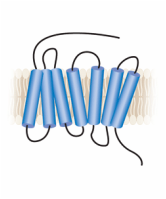
Although a protein may consist of several domains, DRD2 is only characterized by one domain, as indicated by PFAM and SMART--databases used to identify and analyze protein domains. This domain is the '7tm_1' or the seven-transmembrane domain, which weaves in and out of the cell membrane seven times as illustrated in the image to the right: |
Below, the diagram on the left was generated by PFAM and the diagram on the right was generated by SMART.
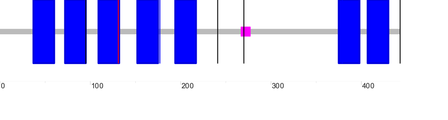
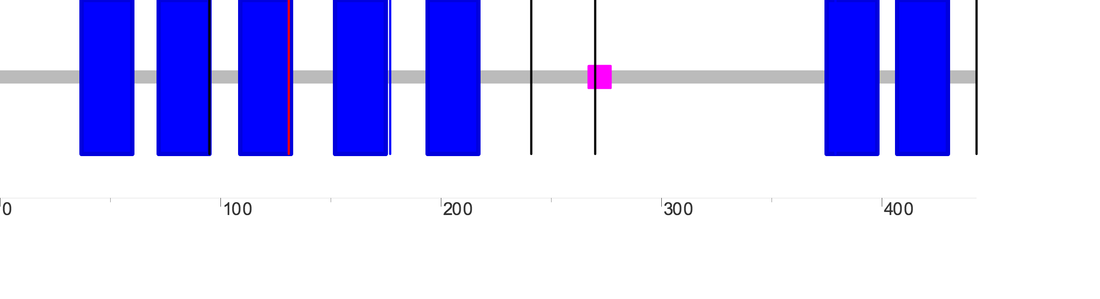
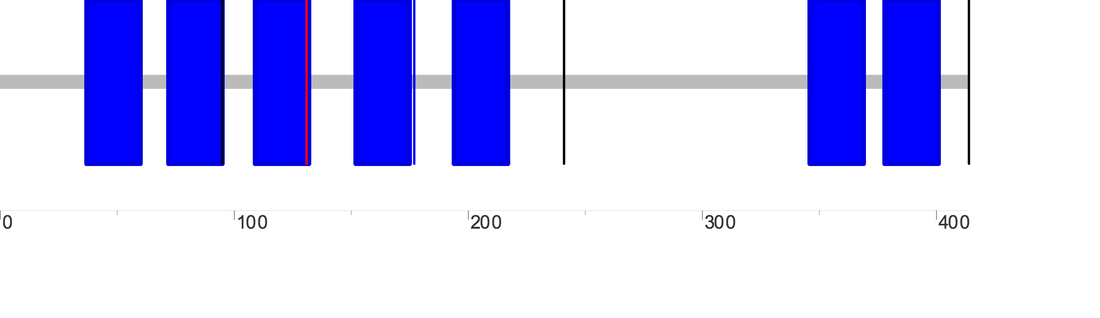
There are two major DRD2 isoforms, and the domain outputs for the long (D2L) and short (D2S) sequences as generated by SMART are as follows (D2L is on the left while D2S is on the right):
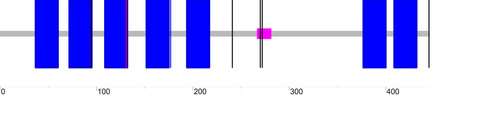
A third longer DRD2 isoform (D2(Longer)) has also been described; its domain output as generated by SMART is as follows:
One blue box represents one section of the transmembrane domain, the pink box represents a low complexity region, while the single vertical lines represent introns. According to SMART, the transmembrane domain for D2L, D2S and D2(Longer) span positions 37-429, 37-400, and 37-431 of the protein chain, respectively. The diagram below is simplified representation of the DRD2 protein isoforms and transmembrane regions (in blue) aligned relative to each other. The numbers on the right tell the total number of amino acids for each isoform.
Discussion_________________________________________________________________________________________________
|
The transmembrane domain tells us that DRD2 is very likely found embedded in the cell membrane. As DRD2 is a dopamine receptor, this makes sense since being positioned at the cell membrane allows interaction and signal transmission with the extracellular environment, to which dopamine is released.
The domain structure in for all three DRD2 isoforms are highly similar, albeit spanning different lengths. All three isoforms retain the domain, with the transmembrane sections starting at approximately the same locations on the protein chain. Information from the structures alone may suggest very little to no differences in function between the DRD2 isoforms. However, according to the SMART diagrams, D2S has a missing intron while and D2(Longer) has an extra line relative to D2L. This indicates alternative splicing, which may mean slight structural differences in each isoform. This can result in very subtle functional differences between each isoform. It is known, for example, that the two extra amino acids in D2(Longer) is in the third cytoplasmic loop, which may possibly affect the binding of intracellular proteins to DRD2 [2]. |
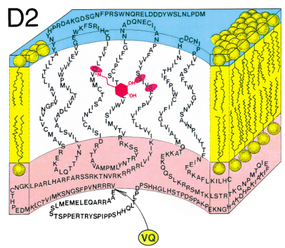
Figure 1. The two extra amino acids, VQ (valine and glutamine), are found in the third cytoplasmic loop of D2(Longer).
Image source: Seeman et al. (2000) [2] |
References
(1) What are protein domains? https://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
(2) Seeman, P., Nam, D., Ulpian, C., Liu, I. S., & Tallerico, T. (2000). New dopamine receptor, D2 Longer, with unique TG splice site, in human brain. Molecular brain research, 76(1), 132-141
(1) What are protein domains? https://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
(2) Seeman, P., Nam, D., Ulpian, C., Liu, I. S., & Tallerico, T. (2000). New dopamine receptor, D2 Longer, with unique TG splice site, in human brain. Molecular brain research, 76(1), 132-141